FDA draft guidance opens pathway for flavoured vapes in US
March 18, 2026

A new draft guidance from the US Food and Drug Administration (FDA) setting out how flavoured vape products could gain regulatory authorisation is being positioned as a potential turning point for the global vaping sector.
The guidance, which outlines evidentiary requirements for Premarket Tobacco Product Applications (PMTAs) for flavoured electronic nicotine delivery systems (ENDS), is the first time the FDA has formally set out a framework under which such products could be assessed for approval.
Crucially, it introduces the concept of Device Access Restrictions (DAR), including biometric authentication, geofencing and continuous age verification, as potential tools to meet the agency’s “appropriate for the protection of public health” (APPH) standard.
Industry observers say the move could open a lawful pathway for flavoured products in the US, a segment that has largely been dominated by illicit or unauthorised devices in recent years.
Vape technology firm Ispire Technology Inc. said the guidance could “unlock” a market worth an estimated $50 billion annually, citing data suggesting that as much as 70 per cent of the current US vape market operates outside formal regulatory approval.
The company, which has a 40 per cent stake in compliance technology venture IKE Tech, said the FDA’s recognition of device-level safeguards validates its long-standing focus on age-gating technology embedded within devices.
“We believe this guidance represents a major step toward a technology-enabled regulatory framework for the vapour category,” said Michael Wang, co-chief executive of Ispire.
“FDA’s recognition of Device Access Restrictions validates our long-held position that continuous, device-level age verification can protect youth while preserving adult choice.”
The FDA document also signals a shift away from reliance solely on point-of-sale age checks, noting that traditional retail safeguards may be insufficient on their own to prevent youth access.
Instead, regulators appear increasingly open to systems that restrict usage at the device level, a development that could have far-reaching implications for manufacturers, retailers and compliance models.
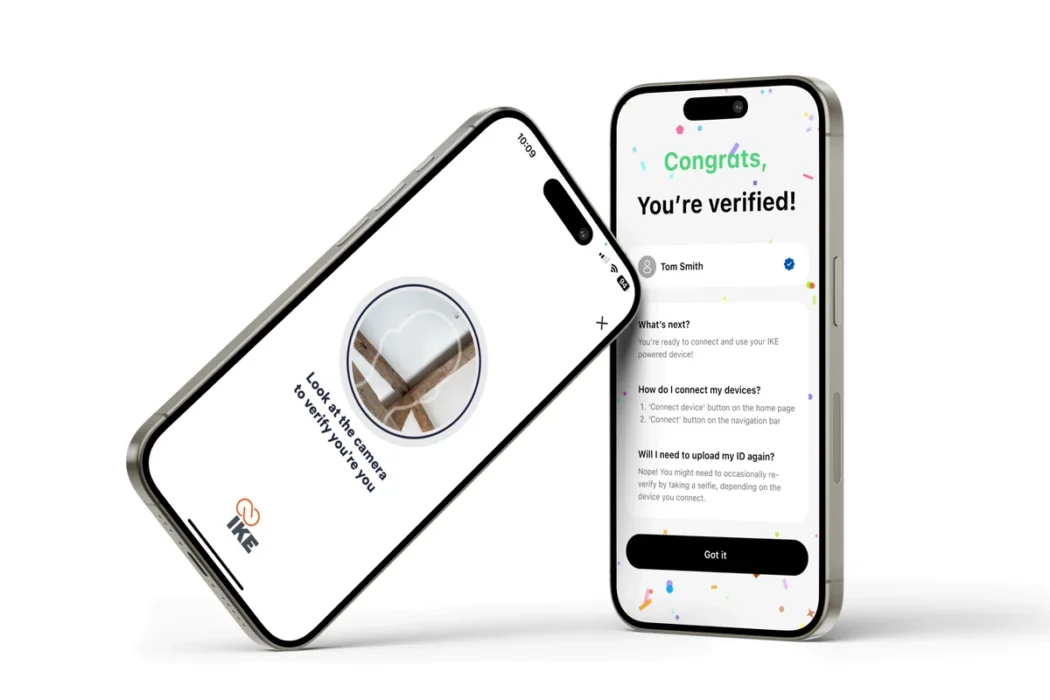
Ispire said its IKE Tech joint venture has already submitted what it claims is the first component PMTA for a standalone age-verification system designed to integrate across ENDS devices. The platform combines Bluetooth-enabled hardware, biometric authentication and identity verification tools to ensure only authorised adult users can activate devices.
According to the company, testing of the system demonstrated full effectiveness in preventing underage access, alongside high levels of user accuracy and satisfaction.
Beyond youth access prevention, the technology is also being positioned as a tool to tackle illicit trade, with capabilities to authenticate products and identify counterfeit devices – an issue that continues to challenge regulators in the US and other markets.
The company added that it is in discussions with major tobacco firms and independent vape brands regarding integration of its technology into future PMTA submissions.
